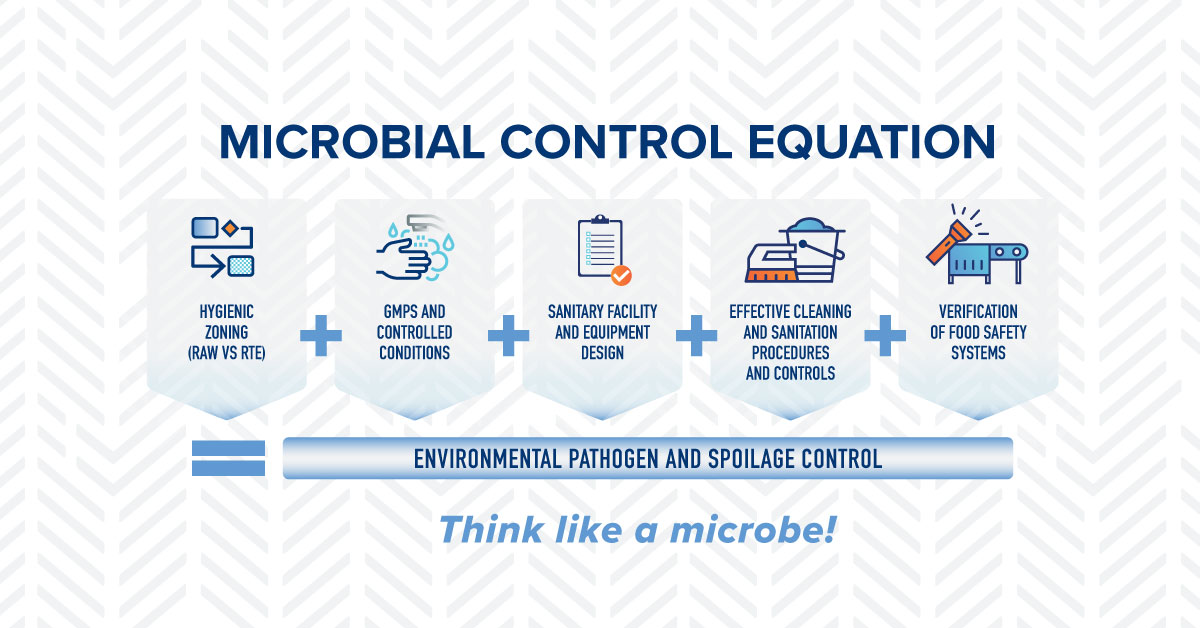
During the month of September, we are celebrating National Food Safety Education Month with a campaign focused on the Microbial Control Equation (MCE). The MCE is a tool that simplifies the different aspects of microbiological control in a food processing and handling environment by combining them into five variables that are easy to remember and communicate. The fourth variable of the MCE is Effective Cleaning and Sanitation Procedures and Controls. This includes the development of effective cleaning procedures based on the 8 Steps of Sanitation in addition to a comprehensive Master Sanitation Schedule (MSS). These tools are widely applied in the food industry, validated by science, and accepted as best practices. Following the 8-Steps of Sanitation and staying up to date on MSS cleaning tasks ensures that the food processes and environment are free of soils and harmful microbes, protecting consumers and your brands.
Cleaning procedures or Sanitation Standard Operating Procedures (SSOPs) are the tools used to document and train on each step of the sanitation process for processing equipment, facility infrastructure, and other tools used in food processing. These procedures should include all the pertinent information needed to communicate how cleaning should be performed, the order of the cleaning process following the 8 steps of sanitation, and the chemicals and tools needed to perform each step. Cleaning procedures should be written specific for the equipment or area being cleaned and include employee safety precautions, personal protective equipment (PPE) needed, as well as relevant food safety information for the tasks being performed. The procedures should be validated to demonstrate that they consistently achieve the level of cleanliness necessary to produce safe, high quality food products.
PSSI’s 8-Steps of Wet Sanitation provides a stepwise process for consistently achieving microbial control with research to validate effectiveness. During step 1, following shut-down, employees prepare for sanitation by locking out equipment, protecting sensitive equipment, removing product and packaging from the area, disassembling equipment, and removing the bulk of food soils from the equipment and environment to enable an effective and efficient first rinse and prevent drains from backing up. Step 2, the first rinse is critical for the effectiveness and efficiency of the rest of the process. During this step the equipment and area are rinsed from the top down with hot water of sufficient pressure to remove 95% of the soil from surfaces. This enables detergents applied during step 3 to effectively break down the remaining soil for efficient removal.
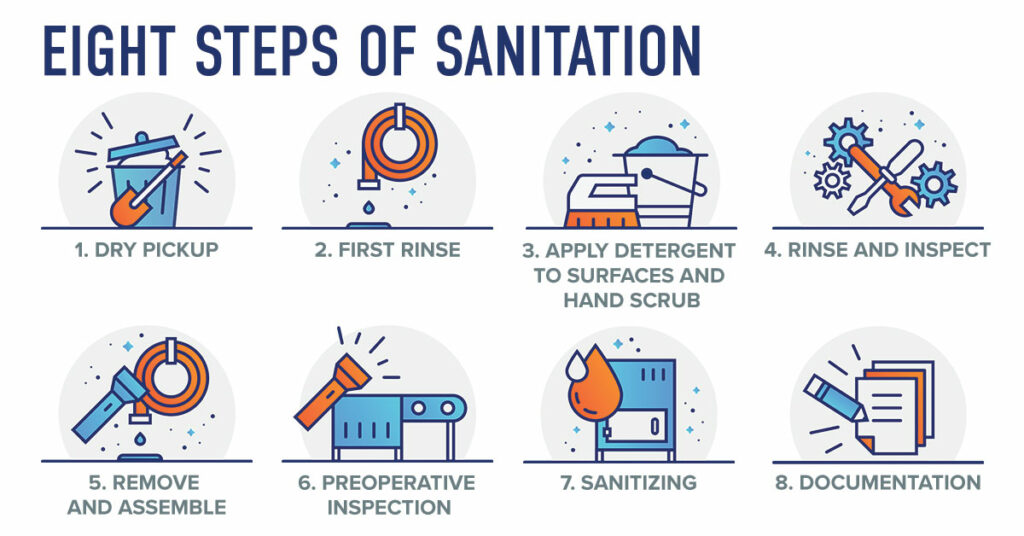
During step 3, detergents are applied, often using foam applicators, starting with the floors, and working upward along the walls and equipment ensuring that all surfaces are coated including the undersides and internal surfaces of the equipment. It is important to verify the four factors of washing during this step including time, temperature, concentration, and mechanical action. This is also the right time to have dedicated drain handling employees clean drains to avoid the risk of cross-contamination. Once detergent is applied, hand scrubbing is performed on difficult to clean surfaces to help break down soils. Then a final rinse is performed from the top down using low pressure high volume hot water prior to allowing detergents to dry on surfaces. Our research demonstrates that when performed correctly, steps 3 and 4 are the most critical for the removal of microorganisms (Figure 1).
Figure 1. E. coli O157:H7 populations during critical steps of the 8 Steps of Sanitation process.
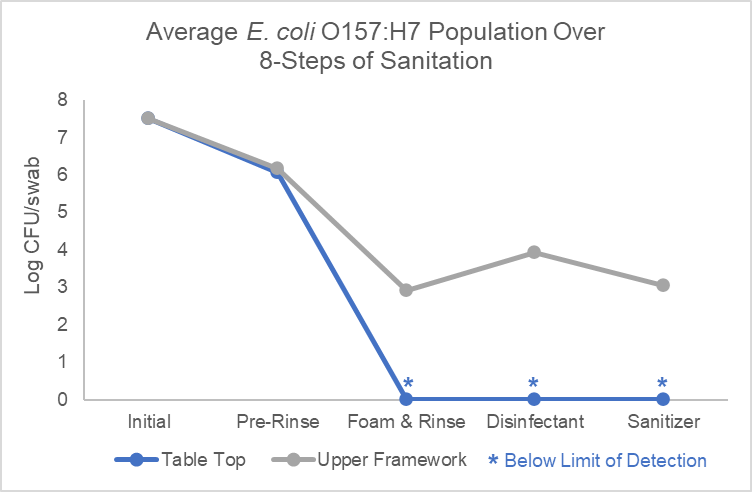
Further, our research also demonstrates that when soil is left on the surface after final rinse it will negatively affect the ability for disinfection and sanitizing in subsequent steps, as demonstrated by the results of the upper framework in Table 1. This emphasizes the importance of pre-op inspection during step 6. During this step, it is imperative to use a flashlight and the senses of sight, touch, and smell to verify complete cleanliness. Often, methods such as ATP testing and microbial indicator organism testing for APC, EB, and Yeast and Molds is also performed to verify the consistent effectiveness of cleaning procedures and establish frequencies for periodic deep cleaning as part of the MSS.
If the process is followed correctly to this point, all soil and microorganisms should be removed, and final sanitizing will act as an insurance policy against the risk of cross-contamination from condensation removal or handling during reassembly. It is important to make sure that final sanitizers are applied to all surfaces of the equipment and infrastructure and that only food contact safe concentrations are left on product contact surfaces. Lastly, during step 8, records are maintained that document compliance with the validated procedures including chemical concentrations, and pre-op inspection performance.
Effective Sanitation Procedures and Controls is a critical aspect of the Microbial Control Equation enabling food processors to maintain equipment and facilities free from soils and harmful microorganisms that negatively impact food safety and quality. When used in conjunction with the other aspects of the MCE, PSSI’s 8 Steps of Sanitation offers a validated process to consistently meet objectives, comply with regulatory requirements, protecting consumers and your brands.